Building muscle in the lab
This provides a potential for treating patients with muscle diseases – and for those who would like to eat meat, but don’t want to kill animals.
ETH Zurich Professor Ori Bar-Nur and his team grow muscle cells in the laboratory. In this case, they are mouse cells, but the researchers are also interested in human and cow cells. Promising applications resonate with both: human muscle tissue cultured in the lab could be used in surgery, while human muscle stem cells could help people with muscle diseases; meanwhile, lab-grown cow muscle tissue could revolutionise the meat industry by enabling the production of meat with no need to slaughter animals.
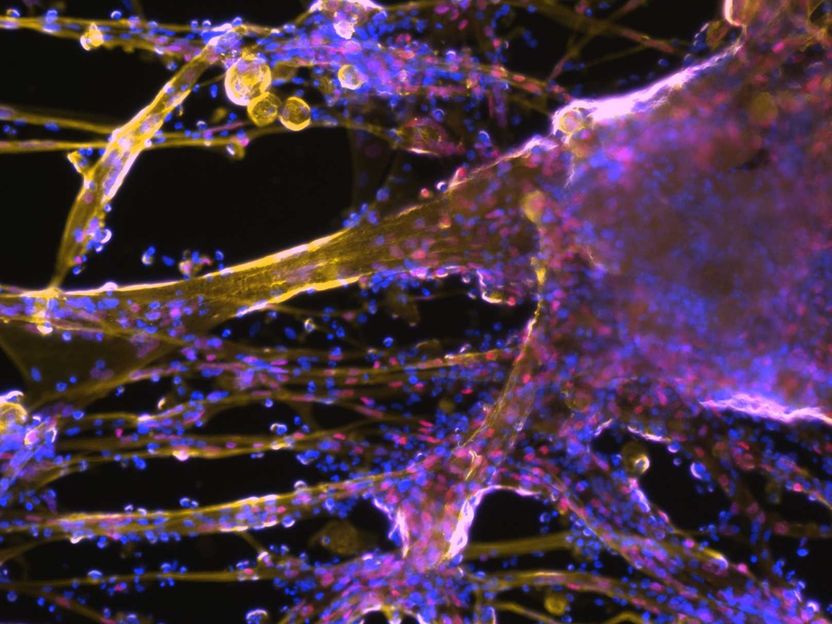
Muscle stem cells and fibers can be grown in the laboratory from reprogrammed connective tissue cells (microscopy image).
Foto: ETH Zürich / Bar-Nur Lab
For now, however, the ETH team’s research is focused on optimising the generation of muscle stem cells and making it safer. They have now succeeded in doing so via a new approach.
Reprogrammed cells
Like other researchers in the field, the scientists at ETH Zurich use a different, easier to grow cell type as the starting material for generating muscle cells: connective tissue cells. Using a cocktail of small molecules and proteins, they molecularly “reprogram” these cells, thereby converting them into muscle stem cells, which then multiply rapidly and produce muscle fibers. “This approach enabled us to produce large quantities of muscle cells,” explains Xhem Qabrati, a doctoral student in the Bar-Nur group and one of two lead co-authors on this study. “Although muscle cells could also be cultured directly from muscle biopsies, the cells tend to lose their functionality after isolation, rendering it challenging to produce large quantities of cells.”
An important component of the used cocktail – and a central catalyst for cell transformation – is the protein MyoD. This is a transcription factor which regulates the activity of certain muscle genes in the cell nucleus. MyoD is not normally present in connective tissue cells. Before these cells can turn into muscle cells, scientists have to coax them to produce MyoD in their nucleus for several days.
No genetic engineering
Until now, researchers have turned to genetic engineering for this process: They used viral particles to carry the DNA blueprint for the MyoD protein into the cell nucleus. There, the viruses insert these building instructions into the genome, enabling the cells to produce the MyoD protein. However, this approach carries a safety risk: scientists cannot control where exactly in the genome viruses insert these instructions. Sometimes the viruses integrate into the middle of a vital gene, damaging it, or this insertion process might lead to changes that can trigger cancer cell formation.
This time, Bar-Nur and his colleagues used a different approach to deliver MyoD to connective tissue cells, inspired by the mRNA vaccines for COVID-19: instead of using viruses to introduce the DNA blueprint of the MyoD gene, they introduce the mRNA transcript of this gene into cells. Since this leaves the cells’ genome unchanged, it avoids the negative consequences associated with such changes. The mRNA still enables the connective tissue cells to produce the MyoD protein, such that – together with the other components of the cocktail optimised by the ETH researchers – they can turn into muscle stem cells and fibers.
The researchers recently published their new approach in the journal external pageNPJ Regenerative Medicinecall_made. They are the first to reprogram connective tissue cells into muscle stem cells without genetic engineering.
Help with muscle distrophy
The muscle cells produced this way are also fully functional, as the researchers have shown in experiments with mice suffering from Duchenne muscular dystrophy. In humans, this rare hereditary disease leaves sufferers lacking a protein necessary for muscle stability, meaning they experience progressive muscle wasting and paralysis.
The ETH Zurich scientists injected non-defective muscle stem cells into the muscles of Duchenne muscular dystrophy mice carrying this defect. They were able to show that the healthy stem cells form repaired muscle fibres in the muscles. “Muscle stem cell transplantation of this sort could be especially helpful for patients with advanced Duchenne, who are already severely affected by muscle atrophy,” explains Inseon Kim, another doctoral student in the Bar-Nur group and a lead co-author on this study. The method is suitable for producing large quantities of muscle stem cells required for this purpose. What’s more, the fact that it does so without genetic engineering and the associated risks makes it attractive for potential future therapeutic use in humans.
Alternative meat production
However, the researchers have yet to adapt their approach to human cells; this is their next step. “In addition, we wish to investigate whether it’s also possible to convert connective tissue cells into muscle cells directly in the body by injecting the MyoD mRNA and the other cocktail components into mice affected by muscle diseases,” Bar-Nur says. This approach, too, might one day help human patients.
Finally, Bar-Nur and his team would like to incorporate their new findings into their ongoing work with cow cells – another research stream of the lab. They hope this method would assist current efforts to culture animal muscle stem cells for cultivated meat production, an alternative method to produce meat for consumption.




















































