Mutation that triggered multicellular life altered protein flexibility
Just as a boat can be driven off course by a log in its path, a single, random mutation can send life in a new direction. That scenario, says University of Oregon biochemist Ken Prehoda, illustrates how a random mutation sparked a huge jump in the evolutionary course of a protein important for the evolution of animals.
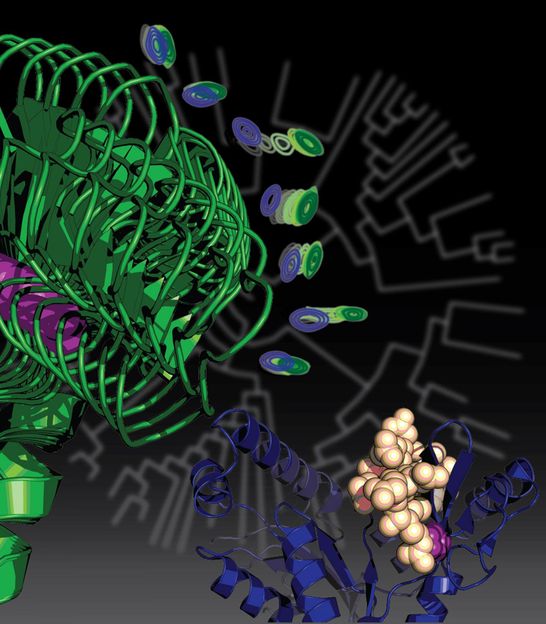
This is an artist rendition of protein evolution showing an evolutionary tree in the background denoting the relationship of genes, and proteins in the foreground affected by mutations in those genes.
Courtesy of Kenneth Prehoda
In January, Prehoda was on a team that found that a random mutation 600 million years ago in a single-celled organism created a new family of proteins that are important for multicellular life. In a new paper, Prehoda and colleagues describe what the mutation did to the original protein, an enzyme known as guanylate kinase.
Mutations happen randomly. Most are bad news. Understanding them better, Prehoda said, could potentially point to new treatments for human diseases such as cancer. Occasionally a mutation is good, helping an organism adapt to environmental changes or advancing overall fitness.
Prehoda's lab initially used a molecular technique called ancestral protein reconstruction. The technique allows researchers to move backward in the evolutionary tree to see molecular changes and infer how proteins performed in the past.
For the new study, Prehoda's lab collaborated with researchers at the Medical College of Wisconsin who studied whether the mutation they had discovered had possibly changed the flexibility of the protein. Next, his team turned to computer simulations in the UO's High Performance Computing Research Core Facility to explore how the altered flexibility they isolated, in turn, led to changes in the protein's interactions.
"We found that this mutation that helped our unicellular ancestor to become multicellular, and ended up leading to an entirely new family of proteins that are specific to animals, did so in a very interesting way," said Prehoda, who is the director of the UO's Institute of Molecular Biology. "Amazingly, this one mutation took a protein that was really flexible -- an important trait for its old job -- and made it much more rigid so it could advance to a new function."
The mutation, which researchers labeled s36P, set off a cascade of events in which guanylate kinase interactions took new routes and evolved into more complex multicellular organisms, Prehoda said. The mutation is still conserved in all animals today, he added.
"A lot of the proteins that do the work in our bodies can be thought of as molecular machines," Prehoda said. "They move in a way that is coordinated with function. Each protein spins in a circle or motors along filaments. Our protein, before the mutation, was an enzyme that had certain flexible movements related to its function. This one mutation fixed the protein's backbone, locking the molecule into a shape that is important for its new function."
Original publication

Get the analytics and lab tech industry in your inbox
By submitting this form you agree that LUMITOS AG will send you the newsletter(s) selected above by email. Your data will not be passed on to third parties. Your data will be stored and processed in accordance with our data protection regulations. LUMITOS may contact you by email for the purpose of advertising or market and opinion surveys. You can revoke your consent at any time without giving reasons to LUMITOS AG, Ernst-Augustin-Str. 2, 12489 Berlin, Germany or by e-mail at revoke@lumitos.com with effect for the future. In addition, each email contains a link to unsubscribe from the corresponding newsletter.