Abbott Introduces New ALK Genetic Test for Specific Form of Non-Small-Cell Lung Cancer
Abbott announced the availability of a new molecular test in the European Union and New Zealand designed to identify the presence of gene rearrangements associated with a specific form of non-small-cell lung cancer (NSCLC). These rearrangements in a gene known as ALK (anaplastic lymphoma kinase) have been implicated in the formation of cancerous tumors in patients with NSCLC.
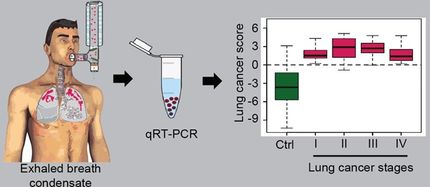
Lung cancer is the world's leading cause of cancer death with more than 1.6 million new cases diagnosed each year. About 85 percent of lung cancer patients have the non-small-cell type and are usually diagnosed with advanced disease with a very low survival rate. About 3-5 percent of those patients are thought to have ALK gene rearrangements and have been shown to respond to therapies that block the ALK pathway. The Vysis ALK Break Apart FISH probe kit, commonly known as the Abbott ALK test, uses Abbott's fluorescence in situ hybridization (FISH) technology to detect rearrangements of the ALK gene on the 2p23 chromosome in tumors. The new Abbott assay will be used for research, testing, and to support evaluations of new therapies in multi-national government funded studies.
Abbott's ALK FISH test was approved in the United States on Aug. 26, 2011. It is commercially available in the U.S. and CE-Mark countries only. Registration of the new test is under way in Australia, China, Korea, Malaysia, Singapore, and Taiwan.
Other news from the department research and development

Get the analytics and lab tech industry in your inbox
By submitting this form you agree that LUMITOS AG will send you the newsletter(s) selected above by email. Your data will not be passed on to third parties. Your data will be stored and processed in accordance with our data protection regulations. LUMITOS may contact you by email for the purpose of advertising or market and opinion surveys. You can revoke your consent at any time without giving reasons to LUMITOS AG, Ernst-Augustin-Str. 2, 12489 Berlin, Germany or by e-mail at revoke@lumitos.com with effect for the future. In addition, each email contains a link to unsubscribe from the corresponding newsletter.